Bringing MIPS to Submission Readiness at Scale.
MY ROLE
Led end-to-end product design to bring Verana's MIPS platform to submission readiness
Partnered with data science, product, and support to ground decisions in actual usage data
Reframed product direction based on user segmentation insights
Shipped the quality dashboards and submission workflow adopted by the primary user segment at launch
4
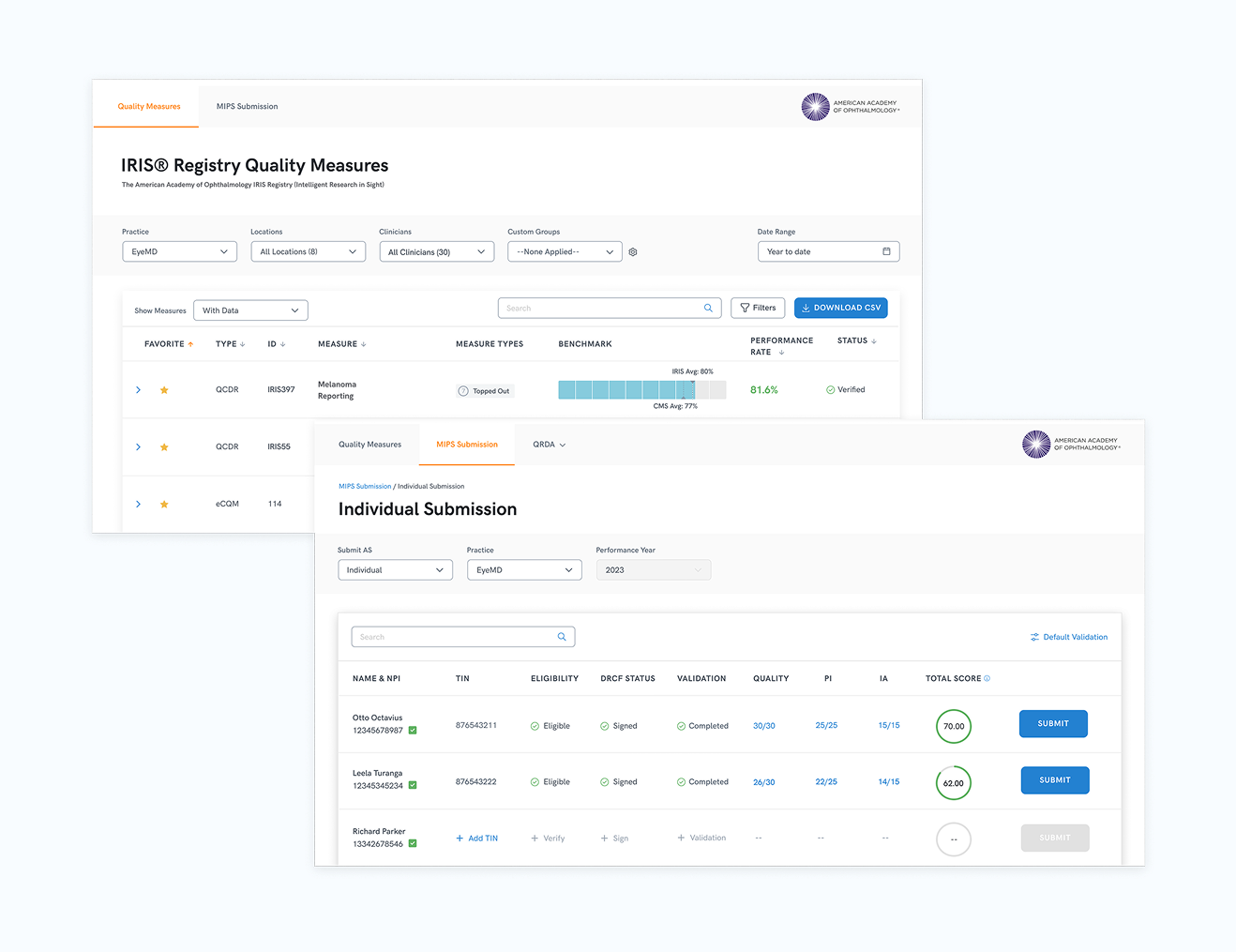
Levels of hierarchy designed to work together: practice, physician, measure, submission
3,000
Clinicians onboarded for first submission cycle
Shipped
Shipped on time and successfully received.
90
NPS at launch. Industry excellent is 50.
THE SITUATION
Every doctor in America has to do this.
Every eligible physician in the U.S. must report performance data that directly impacts Medicare reimbursement.
The process is complex and time-consuming, at exactly the moment clinicians can least afford it. Get it right and you earn a bonus. Get it wrong and you are penalized.
Verana Health, backed by Google Ventures, had built a platform to manage this reporting. I inherited an early version and was tasked with getting it submission-ready before the next cycle.
As the sole designer, I owned the problem end-to-end.
BIG INSIGHT
The product was being built
for the wrong users.
Insight: The loudest voices in the room were not the primary user group.
Academic Medical Centers dominated every internal conversation. They shaped requirements, drove support tickets, and anchored the product's mental model. I asked a data scientist to pull actual usage, because the team's assumptions about who we were designing for had never been validated against the product itself.
Shift: Data showed AMCs were the smallest segment. Private practices were the primary user group by a wide margin.
The two groups wanted fundamentally different things. AMCs cared about quality data and performance tracking. Private practices cared about ease of submission and the confidence that it had gone through correctly. Two different jobs to be done, being served by one product that was pulling toward the smaller one.
Decision: Prioritized the submission workflow over reporting depth, based on actual user segmentation.
This shifted where the design investment went. The dashboard work continued, but the heaviest product effort moved to the submission experience, the surface most private practices depended on daily and the one with the sharpest financial stakes attached to it.

Key decisions that shaped the build
Decision: Submission workflow prioritized over deeper AMC dashboard features
Boundary: Designed for the private practice user, not the institutional researcher
Trade-off: Reporting depth held back to protect submission simplicity
Scope: Rebuilt the four-level hierarchy around the submission task
APPROACH
No users, no research.
An unfinished system.
Direct access to users did not exist when I joined. My prior MIPS experience from Modernizing Medicine let me move fast on domain decisions, but I still needed real signal. I built proxy research from support team data. That produced early decisions grounded in friction, not assumption.
It also earned trust. The support team opened the door to real users once they saw the approach producing results. Those sessions changed specific decisions the proxy research had not surfaced.
THE SOLUTIONReframed the workflow around
submission readiness.
Two constraints I held throughout: status had to be unambiguous at every level, and submissions had to guide users forward without relying on their prior knowledge. The same visual language, the same progression logic, from practice overview to individual submission, designed so the system carried the cognitive burden, not the clinician.
Where it made sense, I added small moments of delight. Not decoration. Interactions that acknowledged effort, confirmed completion, reduced anxiety. Compliance software can be functional and still be something people do not dread opening.
THE OUTCOMEA platform clinicians
actually trusted.
The first submission cycle was the pressure test. 3,000 clinicians moved their reporting through the platform under a regulatory deadline. Support volume stayed low. Private practices, the newly validated primary user segment, adopted the submission workflow and began recommending it to peers.
“People were recommending compliance software to colleagues unprompted. For a category users typically endure rather than enjoy, that was the strongest signal the team could have asked for.”
KEY IMPACTWhat this
changed.
This work delivered impact across multiple areas of the business:
Product
Transformed a complex compliance workflow into a system clinicians could navigate confidently, contributing to a 90 NPS experience at launch
Brand
Shifted the product from something clinicians tolerated to something they actively recommended to peers
Operational
Reduced decision friction and improved submission efficiency, while accelerating development cycles from 4 weeks to 2 weeks through improved cross-functional alignment
User / Market Insight
Revealed that mid-to-large ophthalmology practices, not academic medical centers, were the primary user base, shifting product priorities toward ease of submission
Financial / Strategic
Reduced churn risk by turning a failing product into a reliable compliance platform tied directly to physician reimbursement
Organizational
Built trust across support, data, and engineering teams, enabling direct user access and more informed product decisions
"The regulations don't change, but the users' relationship with those regulations is full of anxiety, confusion, and workarounds. The designer's job is to absorb that complexity so the user doesn't have to."
