SPARC: Shipped 3 Months Early,
Full Scope, 92.4 SUS
MY ROLE
Led end-to-end product design as sole designer
Reframed the brief from systems-first to workflow-first
Established the pattern framework the team built against
Shipped full scope three months early with 92.4 SUS and full adoption
V1.1
Every requested feature delivered plus additional capabilities identified post-launch
3,000
Clinicians onboarded for first submission cycle
3 mo
Ahead of schedule at MVP
92.4
NPS at launch. Industry excellent is 50.
OVERVIEW
Context for non-clinical readers
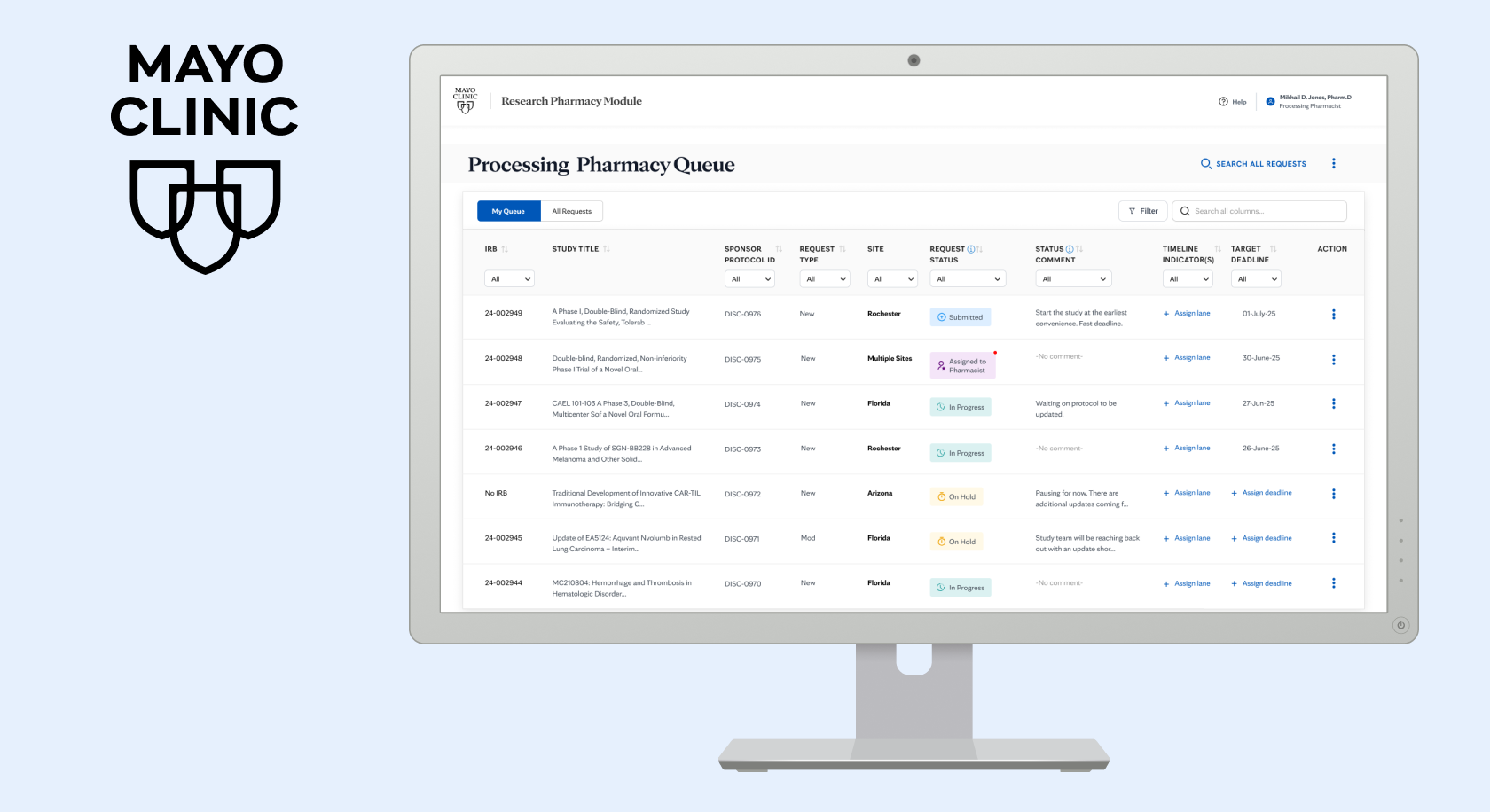
Every clinical trial that involves medication or treatment requires a research pharmacist, a specialist who manages the drugs used in the study, tracks dosing, ensures compliance, and coordinates with the clinical team. At a large academic medical center like Mayo Clinic, one pharmacist might be assigned to dozens of active studies at once, across multiple sites.
When a new study comes in, a Research Protocol Specialist sets it up and hands it off to a pharmacist. That pharmacist may later hand it to another pharmacist. The study might run across multiple Mayo sites simultaneously. At every step, multiple people need to stay in sync without disrupting each other's workflows or losing critical context. SPARC is the platform built to make that coordination visible and seamless.
Reframed the brief.
Bet on the pattern.
Research pharmacists and Research Protocol Specialists at Mayo Clinic needed a shared platform to coordinate clinical trial pharmacy workflows: study intake, assignment, handoff, and multi-site execution. Before SPARC, that coordination lived in email, phone calls, and informal notes, with no shared source of truth.
I was brought in as the sole designer on a project already in motion, with a systems-first brief that prioritized technical architecture over workflow. I reframed the brief around how pharmacists and RPSs actually coordinated, bet the build on a pattern framework rather than screen-by-screen design, and shipped a platform that scored 92.4 SUS, delivered full scope three months ahead of schedule, and reached 100% adoption across all 34 research pharmacists.
The Brief
Systems-first. Technical architecture prioritized over workflow. The project was already in motion when I joined.
The Bet
Pattern framework over per-screen design. The team could build without a designer in every decision.
The Reframe
Grounded the work in how pharmacists and RPSs actually coordinated across the full study lifecycle.
The Result
92.4 SUS. 34 of 34 pharmacists adopted. Shipped three months ahead of schedule at full scope.
THE PROBLEM
Multi-site. Multi-role.
No shared view.
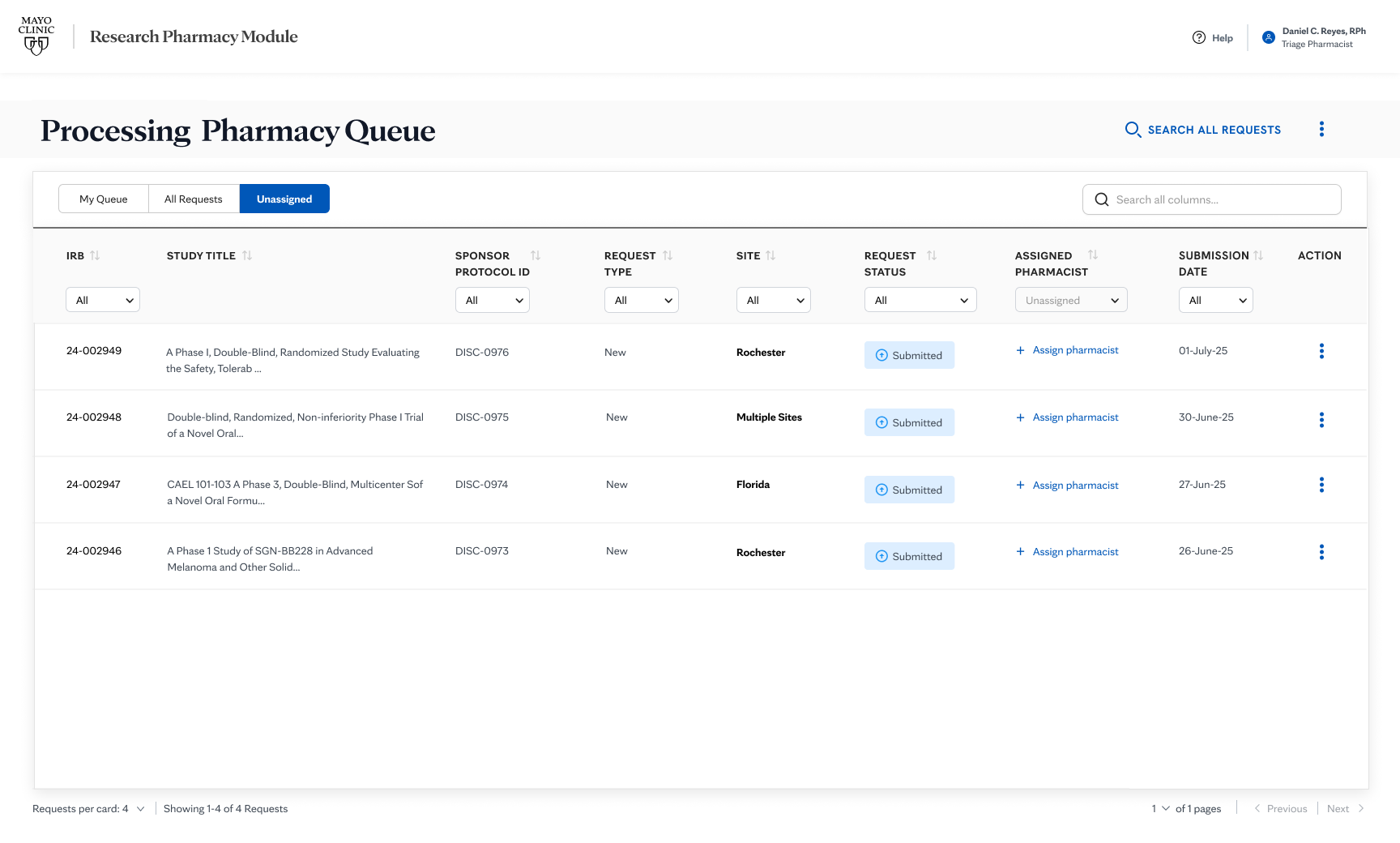
The coordination challenge spanned a full lifecycle and multiple sites. An RPS sets up a study and hands it off. A pharmacist picks it up and may later hand it to a colleague. The study can run simultaneously across Rochester, Arizona, and Florida. At every step, multiple people needed to stay in sync. But the information lived in inboxes and in people's heads instead of a shared surface anyone could see and act on.
THE DECISION
Bet the build on the pattern,
not the screen.
The strategic decision on SPARC was architectural, not cosmetic. A small team was going to build across pharmacist, RPS, and multi-site contexts, each with its own workflow nuances. Designing every screen in isolation would have bottlenecked on me. Designing a pattern framework meant the team could extend the system confidently into contexts I wouldn't be in the room for.
The dev team came in with strong instincts, and those instincts didn't always align with what the research was showing. Rather than relitigating each decision upfront, I invested in documentation: rationale grounded in user research, kept current, visible to the whole team. Documentation became the architecture that held the pattern in place through the build. When UAT came, real pharmacists and RPSs confirmed what the research had indicated. The patterns shipped intact.
Key decisions that shaped the build
Decision
Reframed the brief from systems-first to workflow-first. Grounded the build in how pharmacists and RPSs actually coordinated.
Boundary
Pattern framework over per-screen design. The team could extend the system without a designer in every decision.
Trade-off
Invested in a durable rationale trail rather than upfront argument. Documentation did the work that relitigation would have.
Scope
Full lifecycle coordination: intake, assignment, handoff, multi-site. Multi-site was a day-one constraint, not a v2 feature.
Integration
Patterns built to be extended by the team directly. The framework outlasted the designer being in every meeting.
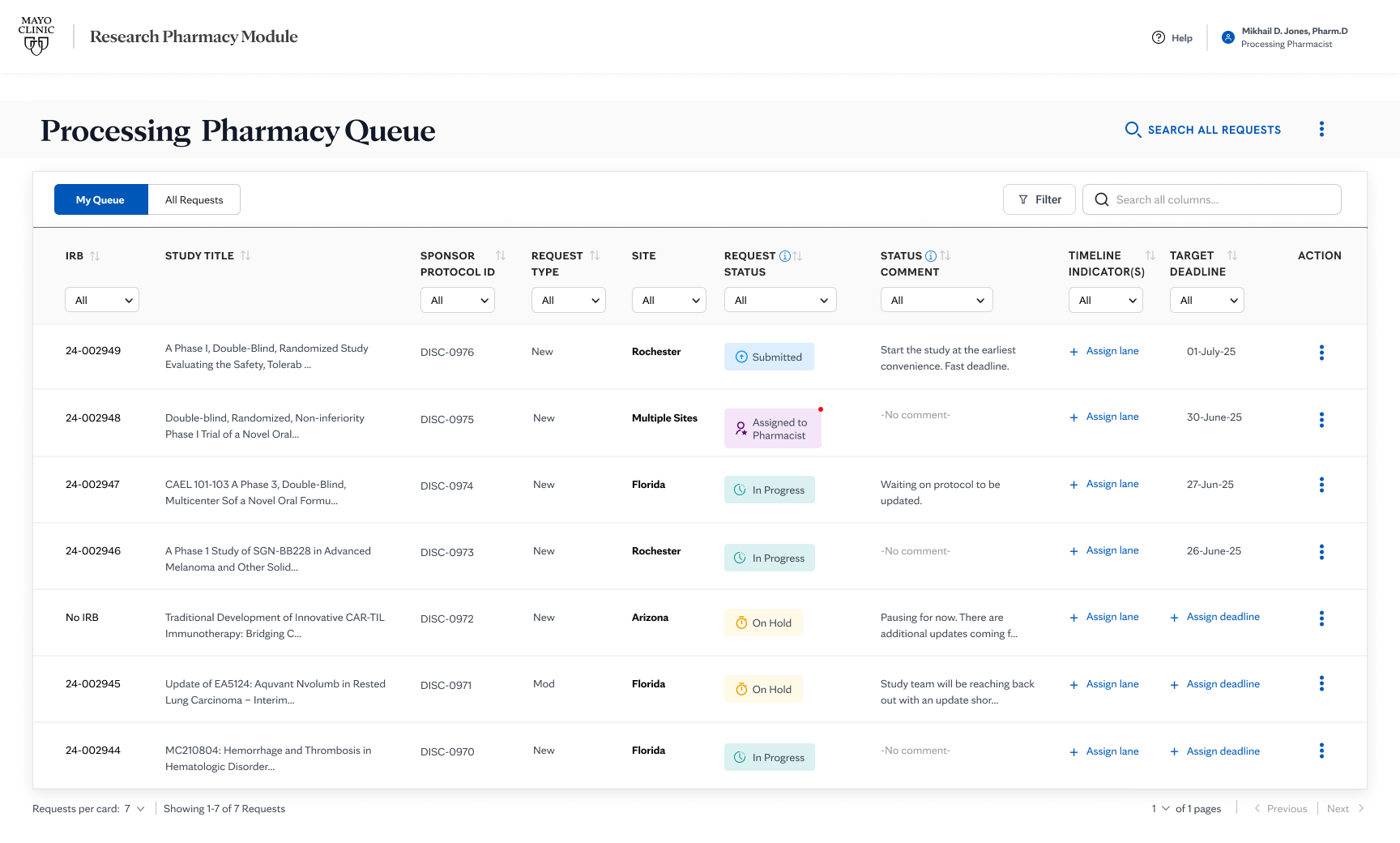
THE SOLUTIONReframed the workflow around
submission readiness.
Two constraints I held throughout: status had to be unambiguous at every level, and submissions had to guide users forward without relying on their prior knowledge. The same visual language, the same progression logic, from practice overview to individual submission, designed so the system carried the cognitive burden, not the clinician.
Where it made sense, I added small moments of delight. Not decoration. Interactions that acknowledged effort, confirmed completion, reduced anxiety. Compliance software can be functional and still be something people do not dread opening.
THE OUTCOMEShipped. Adopted.
Still growing.
The pattern framework did what it was built to do. Once the team had a shared language and a scalable system, the build accelerated. MVP shipped three months ahead of schedule at full scope. Not a trimmed MVP to hit a date, but the complete product before the original deadline.
The 92.4 SUS at launch confirmed the bet: when you design for the actual workflow and give the team a pattern they can extend, pharmacists use the tool without friction. All 34 research pharmacists are active users. By v1.1, every originally requested feature had shipped, plus additional capabilities scoped from real user feedback. The product management team wants to continue building on the foundation.
92.4
SUS score at launch. Industry excellent threshold is 85.
34 of 34
Research pharmacists actively using the platform
v1.1
Second release shipped with features scoped from real user feedback
KEY IMPACTWhat this
changed.
This work delivered impact across multiple areas of the business:
Product
Shipped a scalable pattern framework that held through UAT intact and extended across pharmacist, RPS, and multi-site contexts without rework.
User
100% adoption across all 34 research pharmacists. Coordination that previously lived in inboxes now happens on a shared surface everyone can see and act on.
Strategic
Shifted the team from a systems-first build to a workflow-first one, validated by measurable outcomes. The pattern approach is the foundation for continued investment in the platform.
Organizational
Established user-centered design as a trusted practice with a development team that had not previously worked that way. Subsequent features scope against user research rather than assumption.
SPARC is what happens when you make the design bet early and hold it: pattern over screen, workflow over architecture. The 92.4 SUS, the full adoption, the scope shipped ahead of deadline. That's a framework built to scale past its designer.
